Overview of Incretin and Glucagon Receptor Systems
GLP-1 (glucagon-like peptide-1) is a peptide hormone widely studied in incretin pathway signaling and receptor-mediated cellular processes. In laboratory research, GLP-1 receptor activity is often examined alongside related peptides such as GIP and glucagon to investigate receptor binding, signal transduction, and downstream signaling mechanisms.
GLP-1 receptor signaling is a central area of investigation in peptide receptor biology. Glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon are structurally related peptide hormones that bind to class B G protein-coupled receptors (GPCRs). These receptor systems are extensively examined in molecular research due to their well-defined ligand-binding domains and intracellular signaling mechanisms. Research into GLP-1 receptor signaling and related receptor pathways contributes to foundational understanding of peptide-receptor interactions, second messenger cascades, and comparative receptor selectivity.
Synthetic peptide analogs engineered to interact with these receptor systems are studied in controlled laboratory environments to assess receptor affinity, structural stability, and signaling pathway engagement. Such investigations support the broader scientific understanding of receptor pharmacology and peptide molecular design.
GLP-1 Receptor (GLP-1R) Structure and Ligand Interaction
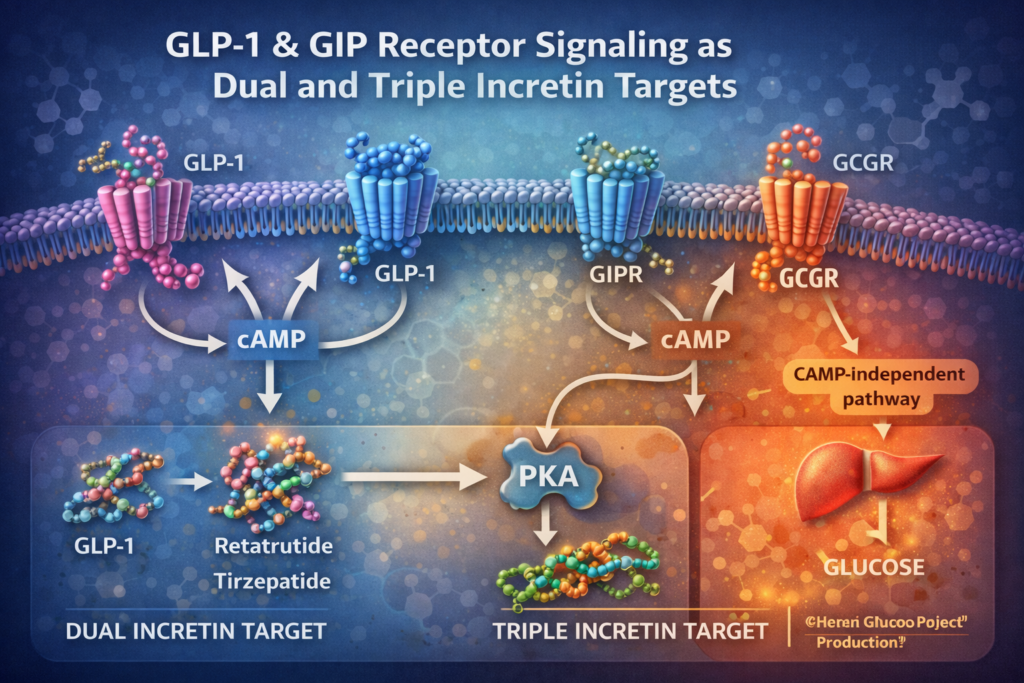
Conceptual illustration of GLP-1 receptor signaling and related GIP and glucagon receptor pathways studied in synthetic peptide research.
The GLP-1 receptor is a class B GPCR that, upon ligand binding, activates intracellular adenylate cyclase via Gs protein coupling, increasing cyclic AMP (cAMP) levels. This mechanism has been characterized through in vitro cell models and structural biology analyses.
Several synthetic peptide analogs referenced in scientific literature for study of GLP-1 receptor signaling include:
- Semaglutide
- Tirzepatide
- Retatrutide
- Mazdutide
- Survodutide
In laboratory research, analysis of these compounds often focuses on receptor binding kinetics, structural stability provided by molecular modifications, peptide half-life adjustments, and conformational behavior in receptor engagement studies. Peptide signaling research also intersects with pathways involved in tissue repair and mitochondrial-derived signaling mechanisms.
GIP Receptor (GIPR) Signaling and Dual-Agonist Peptides
The GIP receptor shares significant structural homology with GLP-1R and signals primarily through Gs-mediated cAMP pathways following ligand attachment. Comparative studies of GIPR and GLP-1R investigate ligand selectivity and cross-receptor interaction dynamics.
Dual-receptor peptide constructs, which permit examination of both GLP-1R and GIPR engagement within a single molecular framework, represent an active area of research. Tirzepatide and Retatrutide are among synthetic peptides cited in literature in relation to multi-receptor interaction profiles. Typical research methods include evaluation of binding selectivity, ligand-receptor conformational analysis, and intracellular response comparisons across engineered models.
Glucagon Recepter (GCGR) and Triple-Receptor Analog Research
The glucagon receptor is another member of the class B GPCR family and similarly signals through Gs protein pathways upon ligand binding. Glucagon receptor studies in research settings often involve structural modeling of receptor domains and examination of ligand activation states using advanced imaging and computational techniques.
Triple-receptor analogs, such as Retatrutide and Survodutide, have been referenced in synthetic peptide literature in the context of their designed ability to engage GLP-1R, GIPR, and GCGR. Laboratory investigations of these analogs frequently explore structural determinants that enable multi-target receptor interaction and comparative receptor binding behaviors.
Amylin Recepter Signaling and Related Peptides
Amylin receptor pathways are studied concurrently with incretin systems due to their interaction with shared metabolic signaling networks. Cagrilintide, described as a synthetic amylin analog in scientific literature, has been examined in receptor-binding studies, structural characterization, and peptide stabilization research. Investigations in this domain often involve evaluation of peptide aggregation properties, receptor complex formation, and the impact of structural modifications on molecular integrity.
Synthetic Peptide Engineering and Stability Modifications
Advances in peptide synthesis and structural modification techniques have facilitated the development of analogs with enhanced stability and receptor engagement profiles suitable for laboratory research. Strategies employed in synthetic peptide design may include:
- Amino acid sequence substitution
- Fatty acid chain attachment
- Backbone modification
- Conformational stabilization approaches
Research exploring these modifications — including studies involving Semaglutide, Mazdutide, and Tirzepatide — contributes to broader scientific understanding of receptor pharmacology, peptide structural behavior, and ligand optimization in controlled research environments.
Conclusion
GLP-1, GIP, glucagon, and amylin receptor systems continue to be active topics in molecular and receptor biology research. Synthetic peptide analogs such as Semaglutide, Tirzepatide, Retatrutide, Mazdutide, Survodutide, and Cagrilintide are referenced in peer-reviewed scientific literature relating to receptor interaction, structural analysis, and multi-target ligand research. Ongoing investigation in laboratory settings supports the advancement of foundational peptide science.
References
- Glucagon-like peptide-1 receptor: mechanisms and advances in signaling pathways. NIH PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC11408715/
- The roles of incretin hormones GIP and GLP-1 in metabolic regulation. NIH PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC12785922/
- GLP-1R signaling and functional molecules in incretin therapy. NIH PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC9866634/
- GLP-1, GIP, and glucagon receptor poly-agonists. NIH PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC9420816/
Related Research Categories
Research materials associated with GLP-1 receptor signaling and incretin pathway models are grouped under: